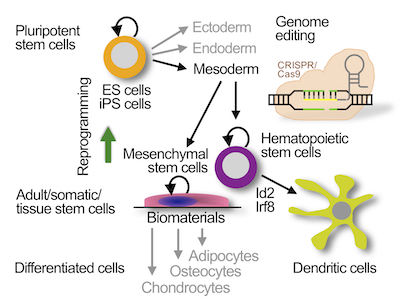
In multicellular organisms, specific stem cell types with distinct developmental potentials occur during development. Transient pluripotent stem cells, which can differentiate into derivatives of all three germ layers (endoderm, ectoderm and mesoderm), are generated during blastocyst development. Adult stem cells, developing at later stages, are more restricted in their potential, since they can differentiate into progenitors and mature effector cell types of only one stem cell system. Adult stem cells have been identified in a variety of tissues in the adult organism and are important for lifelong tissue homeostasis and repair.
Antigen presenting dendritic cells (DC) represent highly specialized immune cells with a central role in immunity and tolerance induction. DC sense antigens, which are taken-up, processed and presented in the context of MHC molecules to elicit antigen specific T cell responses. Specific DC subsets exist that differ in surface phenotype, function, activation state and anatomical localization, including (i) classical DC type 1 and 2 (cDC1 and cDC2, respectively) in lymphoid and non-lymphoid tissues; (ii) plasmacytoid DC (pDC) in blood that represent the major producers of type 1 interferon and (iii) Langerhans cells (LC), the cutaneous contingent of DC in epidermis.